recomWell Borrelia IgG
recomWell Borrelia IgM
Enzyme immunoassay with antigens produced by recombinant techniques for the detection of IgG and IgM antibodies against Borrelia burgdorferi sensu stricto, Borrelia afzelii and Borrelia garinii
The bacterium Borrelia burgdorferi, a spirochete, is responsible for Lyme borreliosis. This disease is transmitted to humans by ticks. Lyme borreliosis is a multisystemic disease with diverse manifestations which make clinical diagnosis difficult. Direct detection of the pathogen is also problematic. Therefore, in routine diagnosis, the serological detection of Borrelia-specific antibodies represents the easiest and safest method for the detection of a Borrelia infection.
Enzyme immunoassays are mainly used for screening. In the recomWell Borrelia tests genetically engineered antigens are used. In contrast to ELISA with lysate antigen from Borrelia burgdorferi cells, the recombinant antigens can be weighted differently when coating, so that otherwise under-represented antigens can be offered in adequate amount. This applies especially to the OspC and the VlsE antigen. In addition, the genetical engineering allows the simultaneous use of antigens from different genospecies.
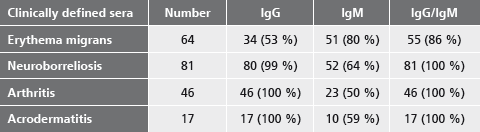
Only the recomWell Borrelia tests detect antibodies against all three genospecies (Borrelia burgdorferi sensu stricto, Borrelia garinii and Borrelia afzelii) safely in one single test run.
Besides the detection of serum antibodies, the recomWell Borrelia tests are ideally suited for the determination of antibodies from CSF. By this the Borrelia specific antibody index from serum and CSF can be calculated, an important criterion of neuroborreliosis.
Product advantages
- Recombinant antigens, therefore
- High sensitivity and specificity
- Optimum presentation without cross-reacting Borrelia proteins
- Excellent discrimination between negative and positive results
- Immunodominant antigens of the three genospecies: B. burgdorferi sensu stricto, B. garinii and B. afzelii
- Use of different immunodominant antigens for the early and late phase:
- IgM: OspC, p41/internal, VlsE
- IgG: p100, OspC, VlsE, p18
- Separate detection of IgG and IgM antibodies
- Identical procedure for IgG and IgM determination
- No RF absorbance necessary in the case of IgM determination
- Easy to quantify
- Standardised CSF-serum-analysis available - CE certified instructions for use
- Easy test procedure; automation possible
- Test procedure and reagents identical in all MIKROGEN ELISA - reagents exchangeable
- Break-a-parts: single sample examination possible
- CE label: The recomWell Borrelia tests meet the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Testprinciple and procedure
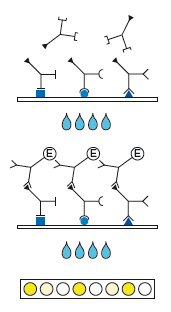 Indirect sandwich test.
Indirect sandwich test.
Recombinant antigens are bound to the solid phase.
1st Incubation
Add patient samples diluted 1:101 (sample: 10 µl of serum or plasma), incubate for 1 h at 37 °C.
Wash 4 times
2nd Incubation
Add peroxidase conjugated anti-human IgG or IgM antibodies (conjugate), incubate for 30 min at 37 °C.
Wash 4 times
Color reaction
Add ready-to-use TMB solution and incubate for 30 min at room temperature. Stop the substrate reaction with H3PO4 and measure the extinction at 450 nm.