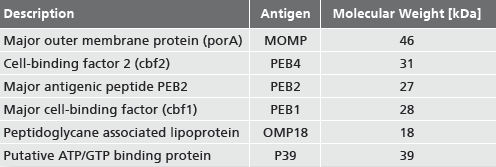
recomLine Campylobacter IgG
recomLine Campylobacter IgA
Strip-Immunoassay with antigens produced by recombinant techniques for the detection of IgG and IgA antibodies against Campylobacter jejuni and Campylobacter coli
The genus Campylobacter comprises gram-negative, spiral-shaped, microaerophilic, mesophilic to thermophilic bacteria with bipolar flagella. Human Campylobacter infections are mainly food associated intestinal infections with world-wide incidence. Contaminated and insufficient cooked foods or drinking water in tropical countries are the main sources of infection. The pathogen reservoir is mainly the intestinal tract of warm-blooded wild, domestic and pet animals. Intestinal Campylobacter infections are the second most frequent enteric bacterial infections reported in Germany after enteric Salmonelloses, whereas unreported cases not reflected in the statistics probably outnumber reported cases many times over.
Campylobacter jejuni is much more frequent, accounting for over 90% of cases as compared to Campylobacter coli at approx. 9%. Besides nearly asymptomatic (clinically inapparent) courses, infected persons suffer from painful gastrointestinal symptoms with sometimes bloody diarrhoea, fever, meningism and myalgias. In rare cases sequelae like postinfectious Reactive Arthritis or Guillain-Barré Syndrome may develop with onset a few weeks after the primary infection.
recomLine Campylobacter is a qualitative in-vitro test for the detection of IgG and IgA antiÂbodies against immunodominant antigens of Campylobacter jejuni and Campylobacter coli. In cases of previous or persistent Campylobacter infection with primary diagnosis based on stool sample culturing, recomLine Campylobacter provides for identification of specific Campylobacter antibodies by means of separate lineups of different antigens produced by recombinant engineering for the purpose of clarifying postinfection complications.
Product advantages
- Recombinant antigens
- High sensitivity and specificity
- Easy and clear interpretation due to easy to read bands
- Easy test procedure; automation possible
- Easy and objective strip interpretation and documentation with recomScan software
- Test procedure and reagents identical in all MIKROGEN strip tests - reagents exchangeable
- Safe evaluation due to strip specific controls (cut-off and conjugate control)
- Separate detection of IgG and IgA antibodies possible
- CE label: The recomLine Campylobacter tests meet the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Testprinciple and procedure
 1st Incubation
1st Incubation
A test strip loaded with Campylobacter antigens is incubated with diluted serum or plasma in a dish for 1 hour.
wash 3 times
2nd Incubation
Peroxidase conjugated anti-human antibodies (IgG or IgA specific) are added. Incubate for 45 minutes.
wash 3 times
Color reaction
5 - 10 minutes after addition of the coloring solution, insoluble colored bands develop at the sites on the test strips occupied by antibodies.
Evaluation
Patient samples from different sources were tested to assess the performance capability of recomLine Campylobacter IgG/IgA. Additional data on the frequency of positive reactivity for the individual recombinant antigens are listed.
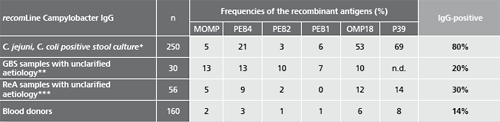
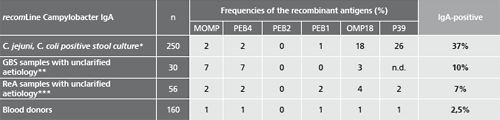
* Samples from patients with positive Campylobacter stool cultures (C. jejuni, C. coli). Blood samples were taken 0 to 40 days after the stool diagnostics, the beginning of the disease was unknown.
** Samples from patients with suspected Guillain-Barré-Syndrom (GBS) unclarified aetiology. No data on previous illnesses involving diarrhoea were known. (Testet with recomLine Campylobacter without P39)
*** Samples from patients with suspected Reactive Arthritis (ReA) unclarified aetiology. No data on previous illnesses involving diarrhoea were known.