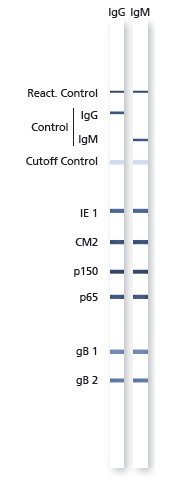
recomLine CMV IgG [Avidity]![]()
![]()
recomLine CMV IgM
Line immunoassay using recombinant antigens to detect IgG and IgM antibodies directed against Cytomegalovirus (CMV) and to determine CMV-IgG avidity Cytomegalovirus (CMV)
Infections with human cytomegalovirus (CMV, HHV5) normally cause mild symptoms, many patients even remain asymptomatically. Immunocompromised patients and pregnant women however are at high risk for suffering damage from a CMV infection. Under immunosuppressive conditions CMV infection or reactivation can cause a severe clinical course of disease. Pregnant women acquiring a primary infection in approximately 40 percent of the cases transmit the virus to the fetus. About 10 percent of the infected newborns show severe damage at birth or are going to develop long-term effects. CMV infection in early pregnancy may lead to abortion or severe clinical symptoms of the affected children, whereas infection in late pregnancy mostly results in mild cases of illness, or even in asymptomatic newborns.
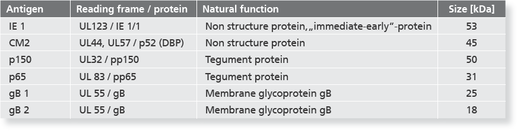
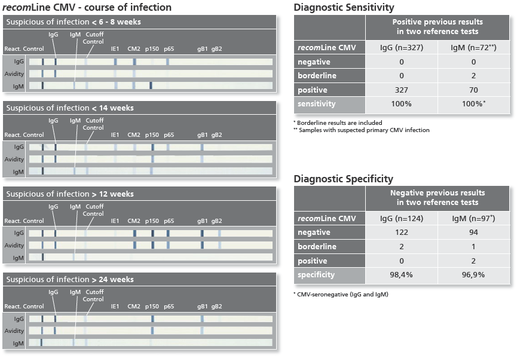
The recomLine CMV is a line immunoassay using all diagnostic relevant CMV antigens in a recombinant format optimized to highest performance in sensitivity and specificity at the same time. Purpose of the assay is the confirmation of positive and unclear screening results (confirmatory assay). Especially the low predictive value of positive IgM results due to persistent or recurrent infections has to be clarified. The test system either is able to determine IgG and IgM antibodies and the avidity of IgG antibodies. Using phase-specific antigens and separate avidity antigens (patented method from MIKROGEN on line immunoassays) the test system can detect CMV specific antibodies in all phases of the infection. Despite viral load the recomLine CMV can be used in pregnancy diagnostic or pre-surgical diagnostic of transplant settings to precisely state if the patient can be judged acute infected or shows typical antigen patterns of a long past infection. While supporting a clear answer to the question what impact the detected CMV antibodies for this particular patient have, this assay can help to optimize therapy, improve disease burden and give patient and doctor relief on reduced acuteness.
Product Advantages
- High sensitivity and specificity ensured by the use of recombinant antigens
- Fast and easy to use due to automation and software based evaluation
- Highly reliable patient management assured by the use of phase-specific antigens which allows to determine the time of infection
- Reliable serological method due to internal cut-off-control and IgG control
- Compatible with all other MIKROGEN line assays - reagents exchangeable
- CE label: The recomLine CMV IgG [Avidity], IgM assay meets the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Test Principle and Procedure
 1st Incubation
1st Incubation
A test strip loaded with CMV antigens is incubated with diluted serum or plasma in a dish for 1 hour.
wash 3 times
2nd Incubation
Peroxidase conjugated anti-human antibodies (IgG or IgM specific) are added. Incubate for 45 minutes.
wash 3 times
Color reaction
8 minutes after addition of the coloring solution, insoluble colored bands develop at the sites on the test strips occupied by antibodies.