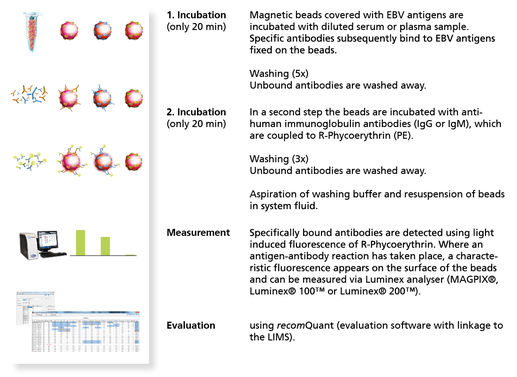
recomBead EBV IgG 2.0
recomBead EBV IgM 2.0
Luminex®-based immunoassay (magnetic beads) using recombinant antigens for detection of IgG or IgM antibodies against Epstein-Barr Virus (EBV) in human serum or Plasma
The Epstein-Barr virus, an ubiquitously occurring herpes virus, can cause the symptoms of infectious mononucleosis (Pfeiffer´s disease) on primary infection. Moreover, as a result of the lifelong persistence of this pathogen, reactivations can occur, especially in immuno-incompetent persons.
Due to the diversity of symptoms caused by EBV infection and their correspondence with the symptoms of other diseases, a secure EBV diagnosis is of great relevance for differential diagnostics. One of the main tasks in routine diagnosis is therefore the serological differentiation of a primary infection from a past infection and the exclusion of an EBV infection.
Product advantages
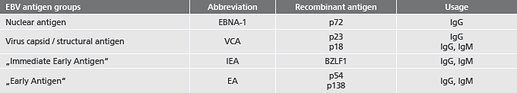
- Reliable screening of EBV by using recombinant Antigens:
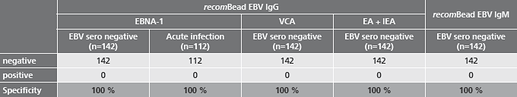
- safe detection of long past infections due to the very high specificity of the diagnostic EBV key antigen EBNA-1
- safe detection of acute EBV infections already in the early phase due to the optimized antigen combination in the recomBead EBV IgG 2.0 and recomBead EBV IgM 2.0 test Systems
- The modern Luminex® multiplex technology integrates the advantages from ELISA (screening) and strip (confirmation) assays:
- fast and easy test procedure similar to ELISA
- fully accomplished for high sample throughput
- only one single test run provides information about the different stages of infection – due to simultanous and seperate detection of EBV specific antibodies
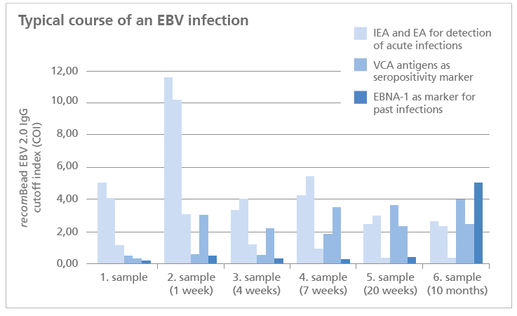
- Safe results:
- evaluation by integrated controls (incubation control, conjugate control, negative control) in each sample
- fully automated processing, analysis and data transfer to the laboratory information system possible
- CE label
Commercial product
| Article number 4554 | recomBead EBV IgG 2.0 (Reagents for 96 determinations) |
| Article number 4555 | recomBead EBV IgM 2.0 (Reagents for 96 determinations) |