recomLine HCV IgG
Strip-Immunoassay with antigens produced by recombinant techniques for the detection of IgG antibodies against Hepatitis C Virus (HCV)
The most important pathogen responsible for parenterally transmitted non-A, non-B hepatitis is the Hepatitis C virus (HCV). This disease is characterized by an incubation period of 2 - 26 weeks and a mild to fulminant course in the acute phase. 50 - 70 % of the patients develop chronic hepatitis which leads to liver cirrhosis in 20 % of the cases. As with hepatitis B, the risk of developing hepatocellular carcinoma is also greatly increased in the case of a chronic HCV infection.
Antibody assays for HCV will be used for the diagnosis of acute or chronic HCV infections as well as for screening of blood donors and blood products.
The recomLine HCV IgG assay is using only recombinant antigens from the region Core, NS3, NS4 and NS5 and allows an easy and a reliable confirmation of HCV screening results.
Product advantages
- Recombinant antigens
- Use of 6 serologically relevant HCV antigens
- Easy and clear interpretation due to easy readable bands
- Control band on every strip
- Very high seroconversion sensitivity for an early stage diagnosis
- 100% sensitivity in confirmed anti-HCV samples
- High specificity for blood donors and clinical samples
- Detection of antibodies for all important HCV genotypes (1 – 6)
- No cross reactivity with other viral hepatitis infections
- Easy test procedure; automation possible
- Easy and objective evaluation and documentation by recomScan software
- CE label: The recomLine HCV IgG meets the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Testprinciple and procedure
 1st Incubation
1st Incubation
A test strip loaded with HCV antigens is incubated with diluted serum or plasma in a dish for 3 hours.
wash 3 times
2nd Incubation
Peroxidase conjugated anti-human antibodies (IgG specific) are added. Incubate for 45 minutes.
wash 3 times
Color reaction
8 minutes after addition of the coloring solution, insoluble colored bands develop at the sites on the test strips occupied by antibodies.
Evaluation
Sensitivity
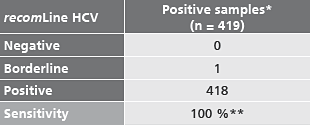
* including samples of genotype 1 -6.
** including of one borderline result
Specificity
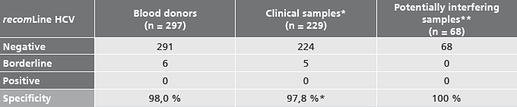
* Samples from patients with other viral hepatitis, recent EBV or CMV infection, autoimmune diseases, HIV, treponema, yellow fever and tick-borne encephalitis infection, pregnant women and samples from the laboratory routine.
** Lipemic, haemolytic and icteric samples, RF-positive samples, patients with hypergammaglobulinemia.



