recomLine HSV-1 & HSV-2 IgG
Strip-Immunoassay with antigens produced by recombinant techniques for the detection and differentiation of IgG antibodies against HSV-1 und HSV-2
The herpes simplex viruses (HSV) belong to the most widespread disease pathogens in the world. There are two serotypes, HSV-1 and HSV-2, which are very similar serologically, however their clinical manifestations are different. In order for an infection to result, direct contact with tissue or secretions containing virus is necessary.
The two serotypes generally prefer different sites of infection, but in principle can infect any area of the skin. In this respect, HSV-1 is primarily responsible for diseases in the area of the head such as herpes labialis (herpes simplex of the lips), while HSV-2 is responsible for diseases in the genital and anal area, such as herpes genitalis (genital herpes) . Severe rare manifestations include, for example, HSV-associated encephalitis or herpes corneae. HSV infection of the newborn (herpes neonatorum), which can occur during or shortly after birth, has particularly severe consequences.
The primary contact with HSV-1 occurs during childhood; that of HSV-2, in contrast, only after the first sexual contact as a young adult [3]. The sero-prevalence increases with increasing age and is markedly higher for HSV-1 that for HSV-2.
Most HSV test kits on the market, including those claiming to detect HSV type-1 or type-2, are based on crude antigen preparations that react equally well with antibodies from HSV-1 and HSV-2 infected patients. As most of the adults already have acquired an HSV-1 infection (seroprevalence is about 83% in Germany) the serological status for HSV-2 can not be reliably proved by crude HSV virus lysate based tests.
The recomLine HSV-1 & HSV-2 IgG uses purified, subtype specific recombinant gG1 (HSV-1) and gG2 (HSV-2) antigens and therefore assures the correct differentiation between an HSV-1 and HSV-2 infection.
Product advantages
- Recombinant antigens, therefore:
- High sensitivity and specificity
- No cross reactions
- Easy test procedure; automation possible
- Easy and objective evaluation and documentation by recomScan software
- Test procedure and reagents identical in all MIKROGEN strip tests - reagents exchangeable
- Safe evaluation due to strip specific controls (cut-off and conjugate control)
- CE label: The recomLine HSV-1 & HSV-2 IgG meets the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Testprinciple and procedure
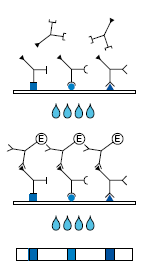 1st Incubation
1st Incubation
A test strip loaded with HSV antigens is incubated with diluted serum or plasma in a dish for 1 hour.
wash 3 times
2nd Incubation
Peroxidase conjugated anti-human antibodies (IgG or IgM specific) are added. Incubate for 45 minutes.
wash 3 times
Color reaction
5 - 10 minutes after addition of the coloring solution, insoluble colored bands develop at the sites on the test strips occupied by antibodies.
Application
- recomLine HSV-1 & HSV-2 as serological confirmation or additional testing:
- For PCR- and/or cell culture findings
- After positive screening with an HSV ELISA
- recomLine HSV-1 & HSV-2 for the determination of the subtype specific HSV serostatus of risk groups:
- Herpes genitalis positive patients and their partners
- Patients with an increased risk for sexully transmitted diseases
- Immunocompromised patients (e.g. HIV seropositive, transplantation patients)
- Recurrend infections in the genital and anal area
- Pregnancy (risk assessment of an primary HSV infection or the threat of an Herpes neonatorum respectively)



