T-Track® SARS-CoV-2
RT-qPCR assay (RUO) for the detection of SARS-CoV-2 specific T-cell reactivity by highly sensitive determination of the relative expression of SARS-CoV-2 antigen-induced specific markers.
Despite the advanced immunization of the population, questions regarding the duration and efficacy of immunization against COVID-19 remain unanswered to date, and the frequency of immunizations still occurs without individualized patient management. The detection of SARS-CoV-2-specific immunoreactivity, ensured on the one hand by the formation of pathogen-specific antibodies (humoral immune response) and on the other hand by a complex system of reactive T-cells (T-cell-mediated immunoreactivity), can help to close these gaps.
T-Track® SARS-CoV-2 is another test among the new RT-qPCR-based assays in our T-Cell Diagnostics product line for the detection of T-cell-mediated immunoreactivity. The test is based on RT-qPCR technique and due to this achieves excellent sensitivity. The use of recombinant antigens and two different markers as read-out make the test different from commonly used SARS-CoV-2-specific T-cell tests. Samples can be collected at several steps during test procedure, providing maximum flexibility and efficiency.
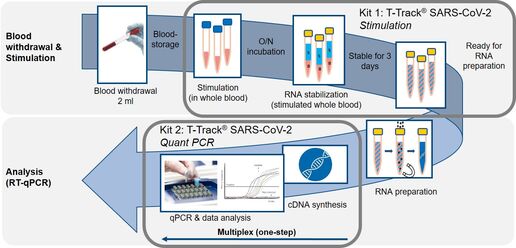
T-Track® SARS-CoV-2 consists of two kits:
1. T-Track® SARS-CoV-2 Stimulation covers stimulation of 2 ml of heparinized whole blood with S1 and NP antigens followed by RNA stabilization.
2. T-Track® SARS-CoV-2 Quant PCR includes the reagents required to perform multiplex RT-qPCR and the evaluation software, which is supplied separately.
Product advantages:
- Highly sensitive RT-qPCR-based T-cell assay (IVD)
- Detection of T-cell reactivity against SARS-CoV-2 antigens (S1 and NP)
- Patented technology with innovative marker combination (IFNG, CXCL10)
- Stimulation in whole blood (2 ml), sample collection during workflow possible
- Semi-automated workflow with software-based analysis


