recomBead Treponema IgG 2.0
recomBead Treponema IgM 2.0
![]()
Treponema pallidum subsp. pallidum is the causative agent of Syphilis, a chronic disease occurring only in humans which is transmitted by sexual contact, via blood or intrauterine. The disease progresses in several stages and starts with a short propagation period at the site of infection (primary stage), followed by the haematogenic spreading of the pathogen throughout the entire organism (secondary stage). After the decline of acute symptoms T. pallidum can persist in the human organism for years (stage of latency) and cause late complications a long time after infection (tertiary Syphilis or neurosyphilis). During pregnancy the pathogen can be transmitted to the fetus (Syphilis connata). An untreated Syphilis can lead to spontaneous abortion, stillborn or premature birth.
For serologic diagnosis of Syphilis a sensitive antibody screening assay (TPHA/TPPA, ELISA) is used in the first step. If this test leads to positive or equivocal results a confirmation assay (FTA-ABS, immunoblot) is performed in the second step to assure the specificity: in the case of TPHA-/TPPA-screening an immunoassay and vice versa in the case of an immunoassay the TPHA-/TPPA-test, alternatives are the FTA-ABS-test or the immunoblot.
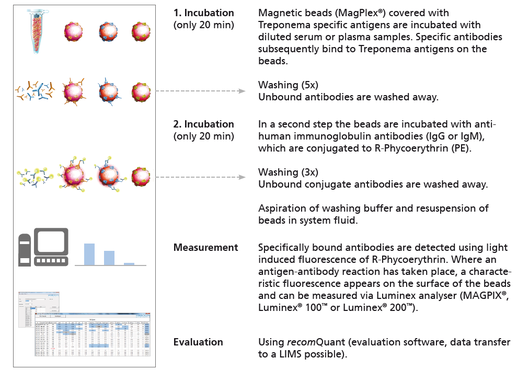
The recomBead Treponema for the detection of IgG or IgM antibodies against Treponema pallidum is based on recombinant antigens and can serve as a confirmation assay for Treponema infections just like the immunoblot.
Product advantages
- Reliable screening with immunodominant, recombinant antigens
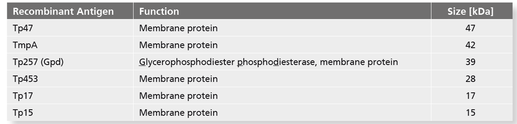
- Use of the highly specific Treponema antigens Tp47, Tp17, Tp15 and TmpA as well as Tp257 (Gdp)
- and Tp453
- Single antigens immobilised on differentiable, colour-coded microparticles (beads)
- Minimal cross-reactivity
- Safe and valid results due to integrated controls
- Incubation/positive control, negative control, conjugate controls
- No additional controls for evaluation and validation necessary
- Easy and comfortable procedure
- High level of automatisation
- Software-based analysis and data transfer to laboratory information systems possible
- Low sample volume sufficient (10 µl)
- Combination of all Mikrogen recomBead 2.0 test systems on one sample plate possible – unique protocol, reagents and test procedure
- Very high accuracy and reproducibility of test results
- Reliable analysis and reporting of follow-up samples
- Quality guarantee by CE-labelling: the recomBead Treponema tests fulfil the high demands of the European Directive 98/79/EC for in vitro diagnostic medical devices.
Commercial product
| Article number 5154 | recomBead Treponema IgG 2.0 (Reagents for 96 determinations) |
| Article number 5155 | recomBead Treponema IgM 2.0 (Reagents for 96 determinations) |