recomWell Yersinia IgG
recomWell Yersinia IgA
recomWell Yersinia IgM
Enzyme immunoassay with recombinant antigens for the detection of IgG, IgA or IgM antibodies against Yersinia enterocolitica and Yersinia pseudotuberculosis
In the past years, Yersinia enterocolitica has become increasingly important as an intestinal bacterium that is pathogenic for human beings. This organism is transmitted orally by contaminated water or food. Typical symptoms of an acute Yersinia infection are diarrhoea, stomach ache, and fever.
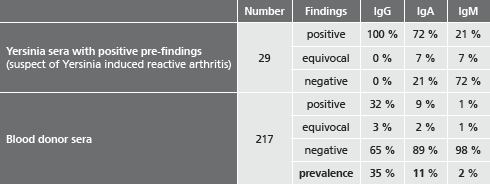
Complications such as reactive arthritis, erythema nodosum and other rheumatic diseases can occur, especially with HLA-B27 carriers. These patients typically show high and persistent IgA titers against Yersinia antigens.
recomWell Yersinia tests are quantitative in vitro tests for the detection and safe identification of antibodies against plasmid-coded virulence proteins. The antigens (YOPs - Yersinia outer membrane proteins) used in these tests are expressed only by strains of Yersinia that are pathogenic for human beings.
The use of recombinant antigens guarantees optimum representation of the immundominant YOPs in the absence of cross-reacting proteins. As a second major advantage, the clarification of Yersinia induced reactive arthritis is supproted by separate detection of antibody classes IgG, IgA and IgM.
Product advantages
- Recombinant antigens, therefore
- Highest sensitivity and specificity
- No cross reactions
- Excellent discrimination between negative and positive results
- Use of recombinant YOPs: All infections with Yersinia pathogenic for man are detected
- Separate detection of IgG, IgA and IgM antibodies possible
- Identical procedure for IgG, IgA and IgM determination
- No RF absorbance necessary in the case of IgM determination
- Easy to quantify
- Easy test procedure; automation possible
- Test procedure and reagents identical in all MIKROGEN ELISA - reagents exchangeable
- Break-a-parts: single simple examination possible
- CE label: The recomWell Yersinia tests meet the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices.
Testprinciple and procedure
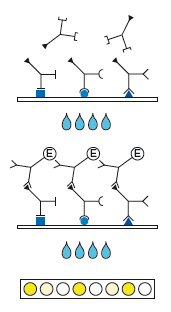 Indirect sandwich test.
Indirect sandwich test.
Recombinant antigens are bound to the solid phase.
1st Incubation
Add patient samples diluted 1:101 (sample: 10 µl of serum or plasma), incubate for 1 h at 37 °C.
Wash 4 times
2nd Incubation
Add peroxidase conjugated anti-human IgG or IgM antibodies (conjugate), incubate for 30 min at 37 °C.
Wash 4 times
Color reaction
Add ready-to-use TMB solution and incubate for 30 min at room temperature. Stop the substrate reaction with H3PO4 and measure the extinction at 450 nm.