recomBead Yersinia IgG 2.0![]()
recomBead Yersinia IgA [IgM] 2.0
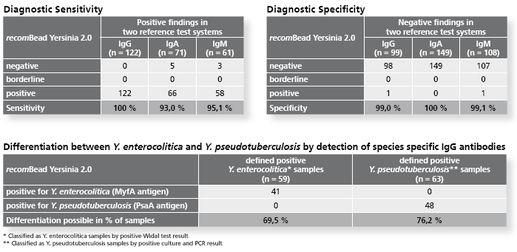
Fluorescence based particle immunoassay using recombinant antigens for the detection of IgG, IgA or IgM antibodies against Y. enterocolitica and Y. pseudotuberculosis. By determination of species specific IgG antibodies, a differentiation between Y. enterocolitica and Y. pseudotuberculosis is possible.
The enteropathogenic Yersinia species, Yersinia enterocolitica and Yersinia pseudotuberculosis, have a global distribution. Transmissions occur orally either in food (especially meat) or in contaminated water. Typical symptoms of an acute Y. enterocolitica infection are watery, sometimes bloody diarrhoea with abdominal pain, vomiting and fever. A Y. pseudotuberculosis infection is difficult to distinguish from appendicitis, it is also referred to as “pseudoappendicitis”. Post infectious complications such as reactive arthritis, erythema nodosum and other rheumatic diseases can occur, especially with HLA-B27 carriers. High and persistent IgA titres against Yersinia antigens are characteristic of these patients.
The modern Luminex® multiplex technology integrates the advantages from ELISA and strip assays of antibodies against individual antigens. Genetically engineered virulence factors and adhesins are used for the multiplex recomBead Yersinia 2.0 test systems. These proteins are expressed only by Yersinia strains that are pathogenic for humans. The recomBead Yersinia 2.0 test systems allow the serologic differentiation of species, as well as the determination of long past Yersinia infections and are thus ideally suited for identification of Yersinia-induced immunopathological complications and chronic yersiniosis. Detection of IgG and IgA antibodies can be a very useful diagnostic tool if Yersinia-induced arthritis is suspected.
Product advantages
- Use of recombinant Yersinia antigens
- Identification of all pathogenic Yersinia by means of Yersinia outer proteins (YOPs)
- Serological differentiation of Y. enterocolitica and Y. pseudotuberculosis infections is possible for the first time with the use of new species-specific Yersinia antigens (PsaA, MyfA)
- No cross reactions with Brucella and other pathogens, as well as no interference caused by LPS
- Fully automated processing and evaluation possible. LIMS interface available.
- Ideal screening or confirmation assay for high sample throughput
- Very high measuring accuracy and very good reproducibility of test results, therefore reliable testing of follow-up samples
- Integrated controls (incubation-/positive control, negative control, conjugate control) - no additional control samples for test evaluation/validation necessary
- Small sample volume (10 μl)
- Combination of all Mikrogen recomBead 2.0 test systems on one plate due to unified processing and exchangeable reagents
- CE label: The recomBead Yersinia 2.0 test systems meet the high standard of the EC directive 98/79/EC on in vitro diagnostic medical devices
Commercial product
| Article number 4654 | recomBead Yersinia IgG 2.0 (Reagents for 96 determinations) |
| Article number 4655 | recomBead Yersinia IgA [IgM] 2.0 (Reagents for 96 determinations) |